The tree of life is often portrayed as a neatly branching structure, with each division point cleanly delineated and separated from its neighbors. The truth is that the various twigs of the tree of life often overlap and become tangled in a process we call symbiosis. I’ve talked about symbiosis before on this blog, which falls along a spectrum of wholesomeness. At one end we have mutualism, a partnership where both organisms benefit and achieve more than the sum of their parts. The other extreme is parasitism, when one organism benefits at the expense of the other. Between the two, there is a broad gray area including commensalism, when one organism’s presence doesn’t necessarily cost or benefit the other in any way. The tree of life is crowded and unpruned, and so sometimes the twigs might wrap around each other quietly and without much fuss. We live on a small planet, and have had to get used to living in uncomfortable intimacy with all sorts of creatures, such as the mites that are living on your eyelashes right now.
But things start to get really weird and tangled when the tree of life loops over on itself twice, or three times, or more. “Three-way” symbioses are surprisingly common, and the more you look for them, the more you realize that the tree of life is more of a knot than anything else.

A recent paper from researchers in Bremen (Germany) and Saudi Arabia looked at such a three-way symbiosis between a coral, a worm and bivalves found off of Tanzania in East Africa. The relationship between solitary corals (Heteropsammia cochlea and Heterocyathus aequicostatus), a sipunculan worm (Aspidosiphon muelleri muelleri) and the clam Jousseaumiella, is a complex triangle of dependencies that had previously been noticed by other researchers, but never investigated at great depth. The worm lives with multiple tiny clams attached, all inside of a small solitary coral the size of a dime (1 cm long). Is the coral a willing host for this crowded boarding house, or has it been parasitized? Does the worm gain anything from the clams? The researchers sought to find out.
Part of the reason I enjoyed reading this study so much was that it had to take a narrative structure to describe the evolutionary ménage à trois of its focus. So much of modern science has moved away from anecdote to hard data, and while there is plenty of that to find in the study, it turns out that a lot of the study of symbiosis is storytelling. We need to know the setting and the characters.
In this case, the main characters are small solitary corals living in the tropical reefs of the Indo-Pacific. We denote them as solitary to distinguish them from their giant colony-forming compatriots that construct the coral reefs currently threatened by climate change and pollution. But like those giant reef-builders, these solitary corals get much of their food from sunlight through a mutualistic partnership with algae called Symbiodinium. The algae provide the host with sugars and other photosynthetic products, and the hosts give them nutrients and a safe cozy home in their tissue.
You might be thinking, “Wait! Dan just said this was a three-way partnership between a coral, a worm and some clams. So this is actually a four-way partnership between corals, worms, clams and algae?” You’d be exactly right. And I’m happy to say that the plot of this sordid story is about to thicken even further.

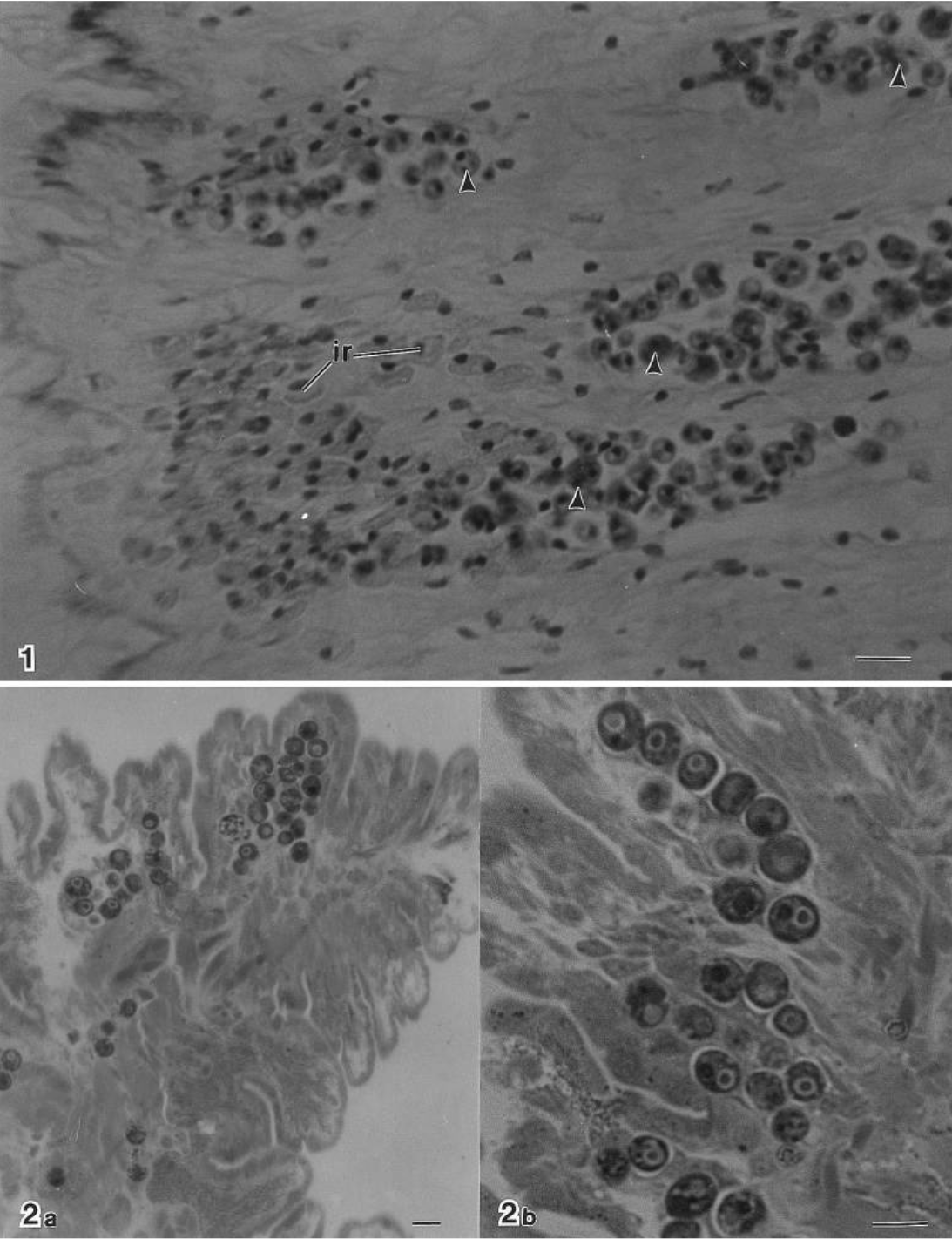
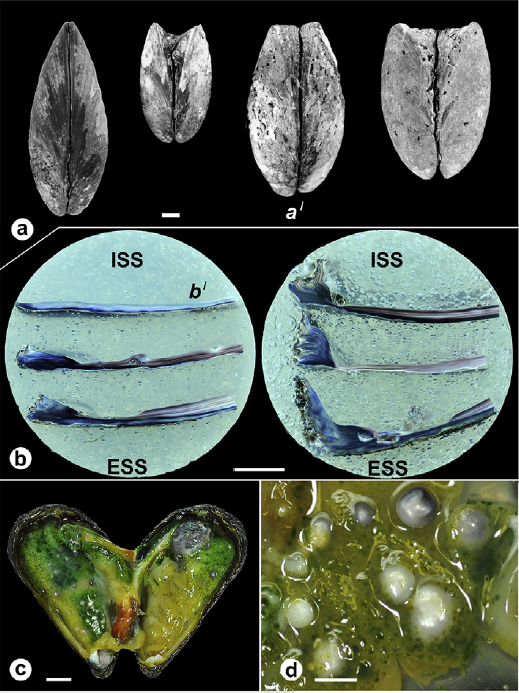
The Aspidosiphon worm is found in a spiral-shaped burrow inside of the skeleton of the coral. It is a pretty cozy home, with walls made of calcium carbonate by the coral, with breathing holes in the sides to allow the worm to breathe and release waste. The researchers wanted to know more about the structure of the burrow. Was it dug out by the worm using acid or an abrasive motion, like some clams use to dig into coral? So the researchers essentially gave the coral a CT scan to see its 3D internal structure. Inside they found growth features suggesting the coral grew around the worm, as if intentionally providing it a home.

Even more crazily, they found evidence that the worm had first settled inside an empty snail shell, like a hermit crab! The coral probably settled on a snail shell as a larva, and grew to engulf the whole snail shell, leaving growing space for the worm inside, with windows and all! So to review, this is now a five-way symbiosis between a dead snail, a worm that moved into its empty shell, the coral (powered by algae) that grew around it and encased the snail shell within its skeleton, and we haven’t even gotten to the clams. How many creatures are hiding stacked in this trench coat? Please bear with me as I explain!

What are the clams doing in this picture? Jousseaumiella is part of a family of clams called Galeommatidae, which we previously mentioned on this blog in the context of some bivalves found growing in the gills of unfortunate sand crabs. Many members of the Galeommatidae family are parasitic or commensal with other marine organisms. In this case, Jousseaumiella are tiny flat-bodied clams less than 1 mm long, found attached to the body of the worm, squeezed inside the burrow in the coral’s skeleton. It feeds on the worm’s waste and potentially food particles coming through the pores in the sides of the burrow. Not the most dignified existence, but a more mobile home means more opportunities to eat a varied diet similar to that that the worm and coral are seeking out, and the clam also gets protection from predation tucked inside the coral. It is unclear if it benefits the worm directly to have clams attached to it.
It is, however, clear how living inside a coral would be a pretty good deal for the worm, which gets a stable, protective suit of carbonate armor to protect it from predators, and grows to fit it as it gets larger. They are normally found inside of rocks, shells and other hard inanimate objects, but having a living home is a cool upgrade. What is the coral getting out of the deal? The researchers note that the corals are often found in the crevices between other large reef-building corals, in areas of the reef that receive high supplies of nutrients and turbidity (dirt that blocks out light). These sorts of environments aren’t necessarily friendly places for a coral to be, since they reduce the light and therefore the food that the coral can receive from photosynthesis. These crevices also have a lot of variability in other conditions like temperature and water flow. But because the coral has hitched a ride on the back of a worm, it can actually move in the sediment to react to changing conditions and avoid being buried by piles of sediment floating by! The worm can also act as a sort of anchor preventing the worm from sinking in the sediment underneath, which would be a big hazard for the small, stubby coral on its own. The coral seems to go to great pains to make its partner comfortable, not growing its skeleton to cover the pore windows to the outside. The researchers note that as coral reefs worldwide are subject to increasing human-made pollution and climate change, it would be interesting to research whether this complex three-(five?) way symbiosis provides the various participants with an advantage compared to other corals.
So like any good story, this symbiosis features complex, growing characters, a dynamic setting, and still plenty of mystery demanding a sequel! To that end, there are lots of other great three-way symbioses to investigate. Snails which farm fungus that parasitizes plants. Bryozoans living on snail shells that have a hermit crab inside. Gobies serving as lookouts at the entrances of burrows built by shrimp, with a crab freeloader along for good measure. Algae and bacteria teaming up to attack mussels. The list keeps going! I could see this becoming quite a franchise!