When I was a student in grade school biology, before I had an inkling of my destiny to become a clam man, I remember learning about food webs. We learned that one part of the food web was made up of autotrophs (“self feeders”), such as plants and algae, which make their own food via photosynthesis (note that there are autotrophic organisms also using chemosynthesis to make food, but we’ll leave those for another blog). The other main portion of the food web are the heterotrophs (“different feeders”), which make their living eating plants, other heterotrophs, or all of the above. The heterotrophs release nutrients as waste which are taken up again by the autotrophs to start the cycle again. It was all so clear-cut and satisfying!
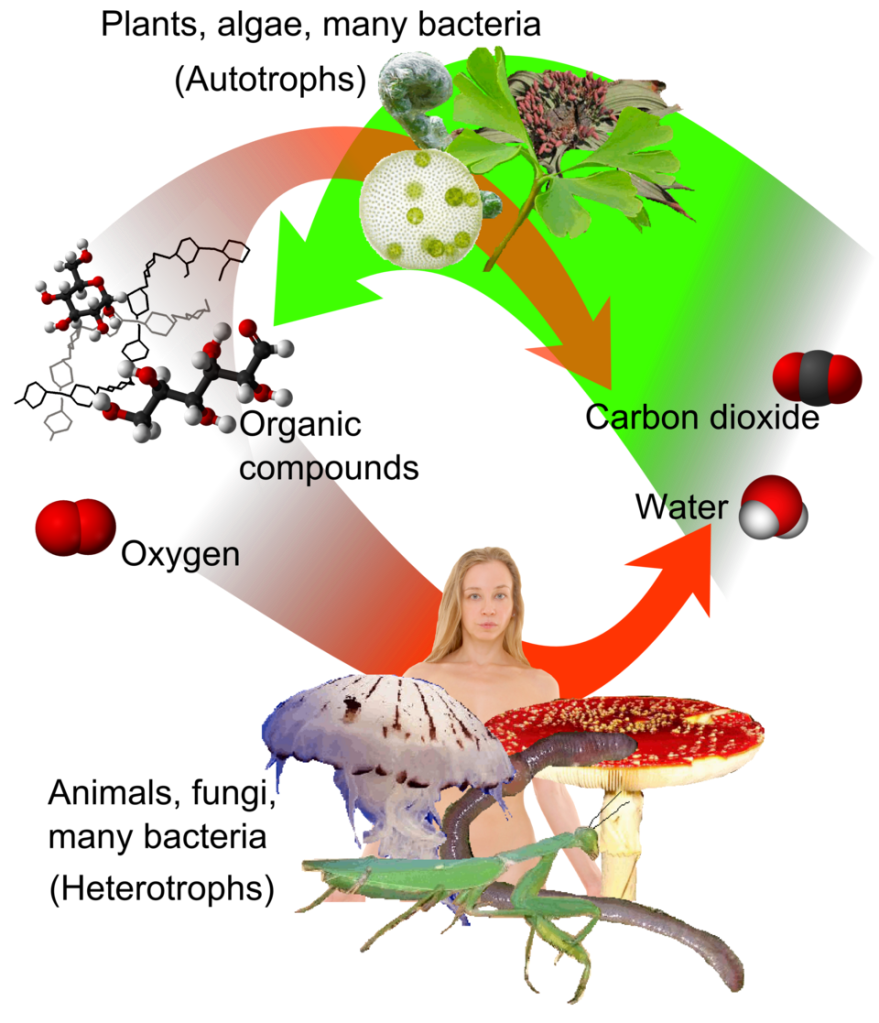
A lot of the tree of life does indeed follow these neat categories, so it made sense for my high school bio class to start with the basics. But at the time, I didn’t understand how many rule-breakers there are, who don’t fit in one neat trophic category. It turns out that between autotrophs and heterotrophs, there is a broad spectrum of weirdos that do a bit of both. We call these weirdos mixotrophs, because they mix different modes of feeding! I have spent a lot of my career studying mixotrophs, not really on purpose, but for two reasons: 1) They’re interesting as heck and 2) Because we don’t know enough about how they work, despite how important they are.
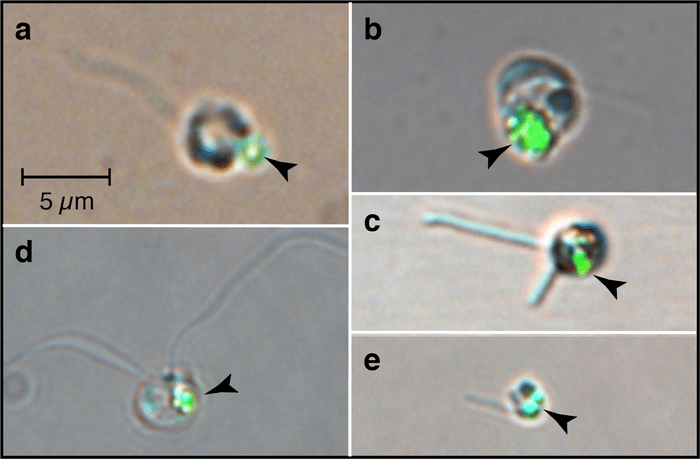
In the ocean, phytoplankton are by far the biggest autotrophic group in terms of biomass. Phytoplankton are tiny algae that float along with currents, using chloroplasts in their cells to gather sunlight, which they use to make sugars through photosynthesis. Those sugars are their food. Most phytoplankton are happy to stick to this autotrophic lifestyle. But a subset of mixotrophic phytoplankton have evolved to not just use sunlight to gain their energy, and instead gain an advantage from munching bacteria or other phytoplankton, using those nutrients as fertilizer for their photosynthesis. Others only are photosynthetic through thievery, stealing chloroplasts from other cells that they harness for photosynthesis, in a process called kleptoplasty. Mixotrophic phytoplankton are able to subsist at times of year when other fully photosynthetic plankton struggle, such as when nutrients or light levels are low. Many of them are able to pursue more active lifestyles through the additional nutrition they gain via their murderous side hustles, allowing them to swim to stay in the highest, brightest part of the water column. This means, somewhat counterintuitively, they can improve their photosynthesis by picking up a hobby of heterotrophy!
It turns out that many types of harmful algal bloom forming phytoplankton are mixotrophs, including Heterosigma akashiwo, which formed a red tide that devastated San Francisco Bay in 2022, killing many kinds of fish. Heterosigma is a champion bacteria eater, and my newest paper investigates the seasonal abundance patterns of it and other harmful plankton in SF Bay. We found a group of several harmful mixotrophic plankton tend to thrive in fall, when their ability to harvest alternative sources of nutrients and swim might give them an advantage over their merely autotrophic counterparts.

It’s not just single-celled algae getting in on the mixotrophy game. Some plants have also seen the benefits of this lifestyle. Some of you may know that I am a bit of a fiend for growing carnivorous plants. I have a small “savage garden” of Nepenthes and Sarracenia pitcher plants, Drosera sundews, Pinguicula butterworts, and of course, the famous Venus flytrap (Dionaea), which Darwin (a huge carnivorous plant fan himself) called “one of the most wonderful plants in the world.” All of these plants are adapted to inhabit nutrient-poor environments like bogs, or high in trees, using specialized leaf traps to catch and digest bugs as a source of nutrients. Some have evolved to make a cozy home for bats that roost and poop in the plant, supplying fertilizer. Nepenthes lowii has evolved to suspiciously resemble a toilet, with a “lid” that attracts shrews to eat tasty nectar, who then poop in the bowl-shaped leaf below. Carnivorous plants are hardcore adapted for the mixotrophy game, to the extent that most have lost the ability to gather nutrients through their roots like other plants. All carnivorous plant lovers learn (sometimes the hard way) that there is no easier way to kill your Venus flytrap than applying Miracle Grow to the soil! They’re poisoned by it!

Even outside nutrient-poor environments, mixotrophic plants are quite common. “Hemiparasitic” plants make part of their living from photosynthesis, but also hedge their bets by using their roots to attack other plants around them. Some, like Indian paintbrushes, consume the symbiotic fungi of other plants’ roots. Others, like mistletoe, live up in the branches of trees, using special stabby needle-like roots to steal nutrients from their host tree. There is a reason that mistletoe became the evergreen symbol of winter love. While their host trees are often deciduous, saving energy by dropping leaves in the light-poor winter months, mistletoe stays merrily green all year, since it just sucks up nutrients from its host tree in the winter!

Many mixotrophic algae and carnivorous plants all have evolved to drop a certain enzyme, NDH, normally found in photosynthetic organisms. Photosynthesis is actually often a pretty stressful process for plants, producing oxidative compounds called free radicals that can cause cell damage. NDH helps control those oxidative compounds, but since carnivorous plants and algae use that oxygen for their increased respiration, like us purely heterotrophic animals do, they often drop the NDH enzyme as an early stage of evolving to be mixotrophic. Researchers are increasingly using the presence or absence of NDH as a indicator of plants that might be mixotrophic!

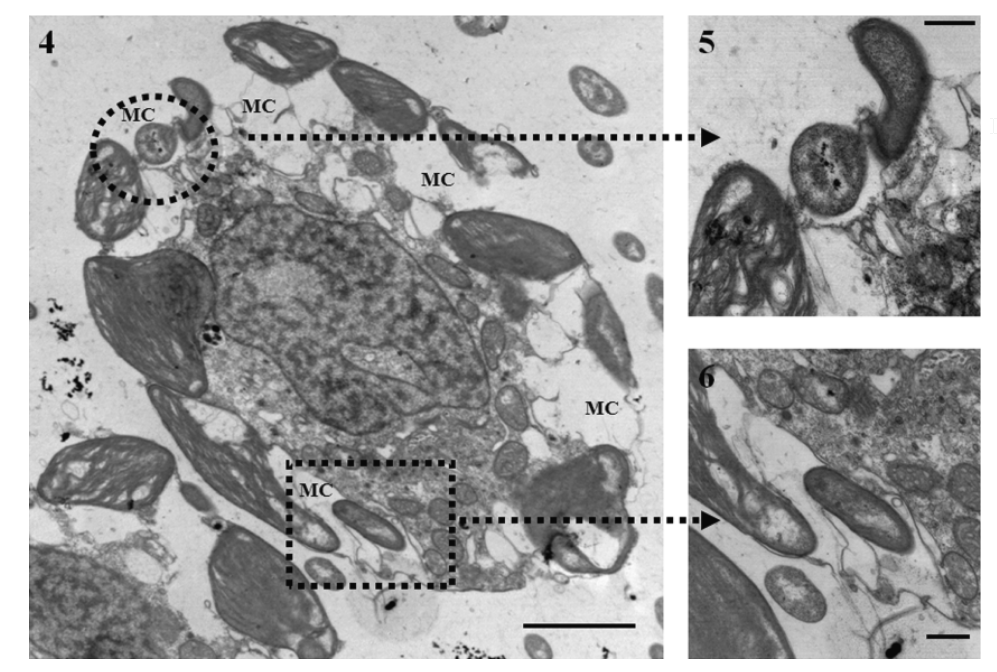
Mixotrophic plants can be a bit murderous, and it turns out that mixotrophic animals can be excellent farmers. Another main direction of my research is studying mixotrophic bivalves, especially the giant clams. Most clams are like cows crossed with vacuum cleaners, making their living filtering and eating phytoplankton from the water. But giant clams have evolved to also partner with the plankton directly in a process called photosymbiosis, pulling some of a type called Symbiodinium into their bodies, where they farm them in networks of tubes in their flesh. This partnership is very successful. By some measures, the photosynthetic efficiency of giant clams is more efficient than some plants! The algae get to eat plentiful nitrogen and phosphorus from the host clam, and they share the sugars they make with the clam in exchange. They even help directly with growing the clam’s giant shells by increasing pH and pumping calcium to their host to use to build the calcium carbonate shell. Mixotrophy is the cheat code that helps giant clams become some of the biggest bivalves the world has ever seen! Their algae help them become giant, and being giant makes them a more effective living solar panel for the algae.

It’s not just giant clams that are mixotrophic. On the coral reefs where giant clams are found, most reef-building corals make use of same photosymbiotic partnership with Symbiodinium. Mixotrophy is the key that makes coral reefs possible, since they are usually nutrient-poor, light-rich places where the algae can’t thrive on their own, but by partnering with the coral, both are able to thrive more than each could on their own! Around the world, mixotrophic animals can be found that have convergently evolved to partner with algae. The vivid green anemones of the tidepools, upside-down jellyfish with their green tentacles, and even some examples of mixotrophic insects and amphibians all point to how when there is a reason, life will evolve outside of the neat boxes of heterotrophy and autotrophy. We keep discovering new species every year that are mixotrophic!

So why isn’t all life mixotrophic? Well, as I mentioned above, it usually is only an advantage in certain environmental conditions. Outside of those specific settings, mixotrophs may be jacks of all trades and masters of none, since they have to maintain all of the cellular machinery needed for both heterotrophy and autotrophy. That is energetically expensive, which can put them at a disadvantage compared to their competitors that specialize in one lifestyle or the other. Giant clams and some other photosynthetic bivalves found today all are in the cockle family, Cardiidae. Recent studies suggest that this family may have unique characteristics in their digestive and immune systems that allow them to host algae in their bodies. Since the host animal is basically taking on a sort of infection of algal cells, they have to make sacrifices regarding aspects of their immune system to keep their symbionts in their bodies. During times when photosynthesis becomes “too” effective, this can backfire. During heat waves, the symbionts of corals and giant clams produce excess oxidative compounds that threaten the host, forcing them to “bleach” and expel their algae to protect themselves.
So at a time when coral reefs around the world are bleaching, under increasing stress from climate change, it is crucial to understand how mixotrophy works. Figuring out the limitations and advantages of mixotrophy might be the key to projecting how corals will respond to future climate change, why some harmful algal blooms happen, and why all sorts of unusual plants can thrive where they do.